Monoclonal antibodies against MERS coronavirus show promise in Phase 1 trial
3 years, 1 month ago
10688
0
Posted on Mar 10, 2021, 7 a.m.
A randomized, placebo-controlled Phase 1 clinical trial of two monoclonal antibodies (mAbs) directed against the coronavirus that causes Middle East respiratory syndrome (MERS) found that they were well-tolerated and generally safe when administered simultaneously to healthy adults. The experimental mAbs, REGN3048, and REGN3051, target the MERS coronavirus (MERS CoV) spike protein used by the virus to attach to and infect target cells. The mAbs were discovered and developed by scientists at the biopharmaceutical company Regeneron, located in Tarrytown, New York. The trial was sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health.
The trial was the first to test the experimental antibodies in people. Conducted at WCCT Global, a clinical trial site in California, the study enrolled 48 healthy adults, 36 of whom received the mAbs. All volunteers were followed for 121 days after receiving mAbs (or placebo) by intravenous infusion. No serious adverse events occurred.
In preclinical studies, investigators at Regeneron and the University of Maryland, College Park, also administered REGN3048 and REGN3051 sequentially and in combination to genetically modified mice that, unlike wild-type mice, can be infected with MERS CoV. When administered one day prior to coronavirus exposure, both REGN3048 and REGN3051 reduced the levels of virus later detected in the lungs, with co-administration providing more potent protective effects than either mAb alone. Similarly, co-administering the mAbs one day after MERS CoV exposure provided a therapeutic benefit in mice by lowering viral levels and lessening tissue damage in the lungs as compared to mice that received placebo.
Together, the findings from the clinical trial and the preclinical mouse studies “demonstrate the potential efficacy and utility of monoclonal antibody therapy for the prevention or treatment of MERS-CoV and lays the groundwork for the development of spike-targeted mAb therapies for other infectious disease threats, including SARS-CoV-2,” which causes COVID-19, the authors conclude.
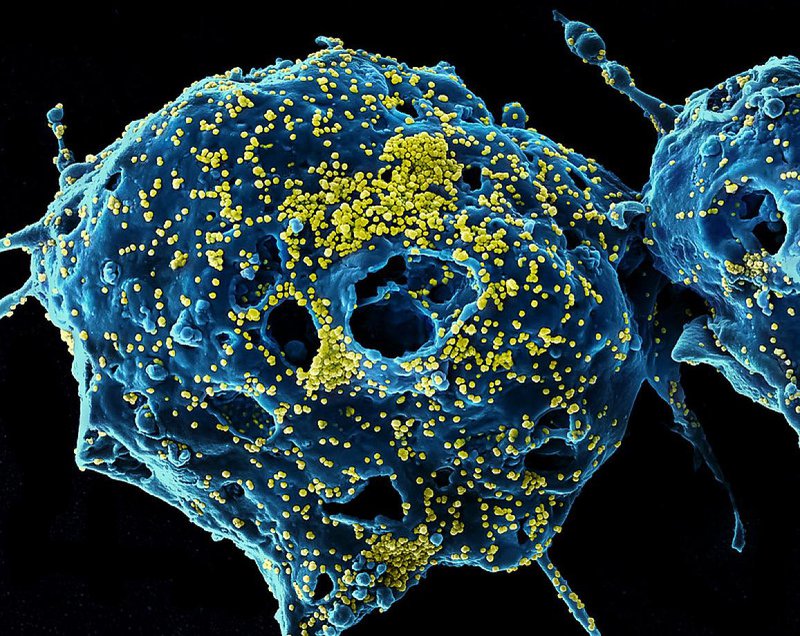
Image: MERS Virus Particles Colorized scanning electron micrograph of Middle East Respiratory Syndrome virus particles (yellow) attached to the surface of an infected VERO E6 cell (blue). Image captured and color-enhanced at the NIAID Integrated Research Facility in Fort Detrick, Maryland.
Image credit: NIAID
As with anything you read on the internet, this article should not be construed as medical advice; please talk to your doctor or primary care provider before making any changes to your wellness routine.
Materials provided by:
Content may be edited for style and length.
This article is not intended to provide medical diagnosis, advice, treatment, or endorsement
Image credit: NIAID
https://www.niaid.nih.gov/news-events/experimental-mers-treatments-enter-clinical-trial
Sivapalasingam et al. Human monoclonal antibody cocktail for the treatment or prophylaxis of Middle East respiratory syndrome coronavirus (MERS-CoV).
The Journal of Infectious Diseases DOI: 10.1093/infdis/jiab036 (2021).









